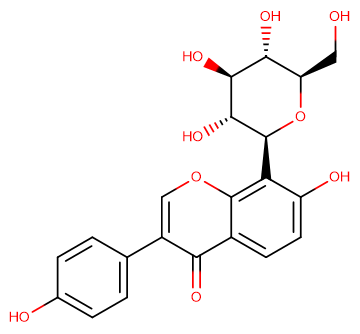
Puerarin
CAS No. 3681-99-0
Puerarin ( Kakonein )
Catalog No. M14266 CAS No. 3681-99-0
Puerarin (Kakonein), an isoflavones found in the root of Radix puerariae, is a 5-HT2C receptor and benzodiazepine site antagonist.
Purity : >98% (HPLC)
 COA
COA
 Datasheet
Datasheet
 HNMR
HNMR
 HPLC
HPLC
 MSDS
MSDS
 Handing Instructions
Handing Instructions
| Size | Price / USD | Stock | Quantity |
| 5MG | 42 | Get Quote |


|
| 10MG | 72 | Get Quote |


|
| 25MG | 147 | Get Quote |


|
| 50MG | 222 | Get Quote |


|
| 100MG | 335 | Get Quote |


|
| 200MG | Get Quote | Get Quote |


|
| 500MG | Get Quote | Get Quote |


|
| 1G | Get Quote | Get Quote |


|
Biological Information
-
Product NamePuerarin
-
NoteResearch use only, not for human use.
-
Brief DescriptionPuerarin (Kakonein), an isoflavones found in the root of Radix puerariae, is a 5-HT2C receptor and benzodiazepine site antagonist.
-
DescriptionPuerarin (Kakonein), an isoflavones found in the root of Radix puerariae, is a 5-HT2C receptor and benzodiazepine site antagonist.
-
In Vitro——
-
In Vivo——
-
SynonymsKakonein
-
PathwayOthers
-
TargetOther Targets
-
RecptorOthers
-
Research AreaOther Indications
-
Indication——
Chemical Information
-
CAS Number3681-99-0
-
Formula Weight416.38
-
Molecular FormulaC21H20O9
-
Purity>98% (HPLC)
-
SolubilityDMSO: 86mg/ml (206.54 mM); Water: <1mg/ml; Ethanol: <1mg/ml
-
SMILESO=C1C(C2=CC=C(O)C=C2)=COC3=C([C@H]4[C@@H]([C@H]([C@@H]([C@@H](CO)O4)O)O)O)C(O)=CC=C13
-
Chemical Name7-hydroxy-3-(4-hydroxyphenyl)-8-((2S,3R,4R,5S,6R)-3,4,5-trihydroxy-6-(hydroxymethyl)tetrahydro-2H-pyran-2-yl)-4H-chromen-4-one
Shipping & Storage Information
-
Storage(-20℃)
-
ShippingWith Ice Pack
-
Stability≥ 2 years
Reference
1.Overstreet DH, et al. Pharmacol Biochem Behav, 2003, 75(3), 619-625.
molnova catalog



related products
-
PF-06424439 methanes...
PF-06424439 methanesulfonate is an oral, potent and selective imidazopyridine diacylglycerol acyltransferase 2 (DGAT2) inhibitor(IC50 of 14 nM).
-
Ac-IETD-pNA
Ac-Ile-Glu-Thr-Asp-pNA is a substrate for caspase-8.Caspase-8 binds to and cleaves the Ile-Glu-Thr-Asp (IETD) peptide sequence to release p-nitroalinide, which can be quantified by colorimetric detection at 405 nm as a measure of enzyme activity.
-
Methyl 2-(Boc-aMino)...
Methyl 2-(Boc-aMino)-2-(oxetan-3-yl)acetate is a chemical compound.



 Cart
Cart
 sales@molnova.com
sales@molnova.com


